Nasal or throat swabs are a thing now! Sticking up a long swap up the nose can be more uncomfortable than it is painful. No wonder why kids and age-old adults freak out! In this case, it is important to make COVID-19 testing as easy and comfortable as possible, at least for the younger and older generations.
Scientists/researchers are scrambling to find additional ways to diagnose the virus to churn out as many tests quickly in a crucial step towards returning to normality. And here’s where Cambridge-based startup Intelligent Fingerprinting becomes relevant.
Easier alternative
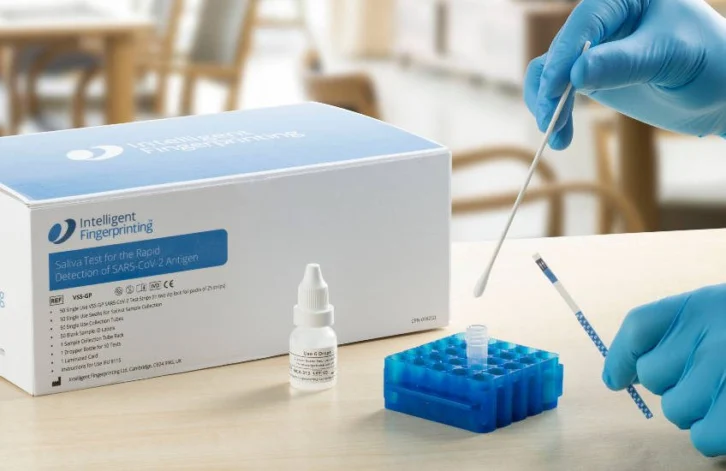
The UK company, which provides fast and cost-effective point-of-care diagnostic solutions, has developed an easy-to-use saliva-based lateral flow test for the rapid detection of the COVID-19 antigen. As per the company’s claims, this test is particularly suitable for more vulnerable people and children.
“Almost everyone who has taken a nose or throat swab to test for COVID-19 knows just how unpleasant they can be. In developing our new COVID-19 saliva test, we have successfully combined the benefits of much simpler sample collection with high levels of accuracy using our existing technology,” explains Intelligent Fingerprinting’s Executive Chairman Philip Hand.
Unlike nose and throat swabs, the company’s test uses a simple swab to collect a saliva sample just from the inside of the cheek. The test results are available in 20 minutes, a bit quicker than the most current nose and throat lateral flow tests.
How does it work?
As explained by the company, a swab, which is deliberately simple in design is used to collect the saliva sample from the inside of the lower cheek. It is then mixed with a reagent buffer that has been pre-added to the collection tube to release the virus.
The screening test is performed by inserting the tip of the test strip into the sample, providing a positive or negative result at 20 minutes.
“The test uses gold immunonanoparticle technology to enable the visual detection of bound antigen via a control line and a red line on the test strip when a saliva sample is positive for the SARS-CoV-2 antigen,” says the company in a press release.
Hand adds, “The introduction of our new COVID-19 test involved rapid design, development, and manufacture within the UK over ten months. Manufacturing capacity at our Cambridge plant, utilising existing equipment and ISO 13485 quality control, is ready to support the production of millions of tests per month, and we also anticipate we will be working with existing and new distribution partners to make this test available across multiple markets,” he added. “At Intelligent Fingerprinting we continue to demonstrate our ability to develop entirely new lateral flow testing concepts, in addition to our unique fingerprint-based drug tests that are sold within the UK, the US, and other international markets.”
Demonstrated 100% sensitivity
Through an independent evaluation, Intelligent Fingerprinting has demonstrated 100% sensitivity in testing 150 positive clinical samples (Ct range 18.0 to 28.0), and 100% specificity following tests with 250 RT-PCR confirmed negative clinical saliva samples.
Now, the company is planning to apply the CE mark on the product under the In Vitro Diagnostics Directive, and also planning to transition to the In Vitro Diagnostics Regulation for European CE marking for professional and self-test use.
Intelligent Fingerprinting: What you need to know?
Founded in 2007 as a spin-out company from the University of East Anglia (UEA), Intelligent Fingerprinting specialises in the development of non-invasive diagnostic screening technology for fast and convenient point-of-care testing using fingerprints.
The company is known for a portable Drug Screening System that works by analysing fingerprint sweat. The company employs around 40 people and has distributors across the globe, including the USA.